FDA AS2 Connectivity
1 Configuring an AS2 connection with FDA
AS2 Gateway fully supports communication with FDA:
- A HTTPS (TLS/SSL) endpoint is available on port 4080, for receiving messages from FDA
- Support for sending messages to FDA over HTTPS, via port 4080
- Full support for standard AS2 features (encryption, signing, compression, asynchronous MDNs etc.)
- Full support for generating and/or configuring custom S/MIME certificates
- Support for custom HTTP headers (FDA routing IDs) for routing messages to appropriate FDA centers
When setting up the connection:
- start by creating a trading station on AS2 Gateway. Configure it to reflect your organization/division (AS2 ID, key pair DN/common name, etc.) - especially the AS2 ID.
during FDA-side configuration (which usually happens via an FDA support ticket), provide the following details to FDA:
- public certificate of above station, for encryption (and signature verification)
- AS2 identifier of the station, as your AS2 identifier and routing ID
- https://service.as2gateway.com:4080/service/as2-receiver as the URL
DST Root CA X3 as the TLS/HTTPS certificate, if requested (this is the root issuer for the TLS cerificate on port 4080, to which FDA will connect to send inbound messages)
All of the above details, can also be found on the partner configuration of your newly created trading station.
- when FDA has confirmed the connection, send a test file to FDA as instructed by them (usually to the
GWTESTcenter).
2 Advanced FDA Integration Support
Additional features to streamline FDA submissions are also available:
Route messages to multiple FDA centers using the same AS2 ID (partner definition).
Define message send profiles that correspond to your frequently used FDA centers, and pick the correct center when sending the message, to automatically add the routing IDs to the FDA-bound message. For example, you can configure the routing IDs FDA center ‘X-Cyclone-Metadata-FdaCenter’ and submission type ‘X-Cyclone-Metadata-FdaSubmissionType’:
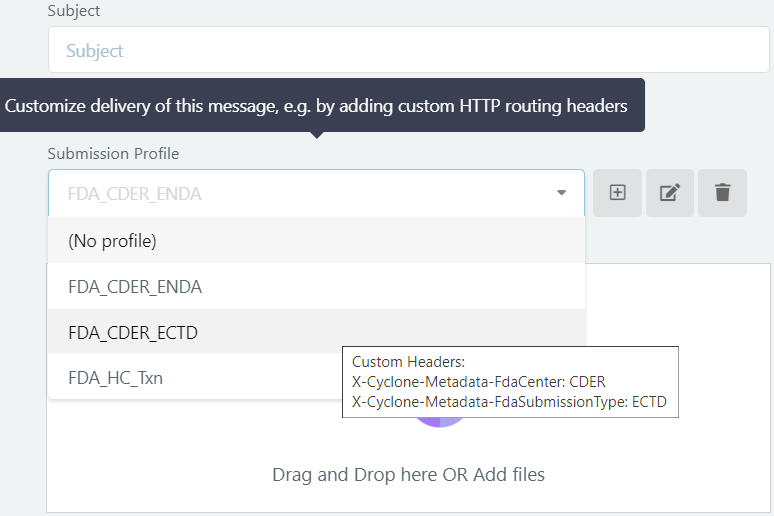
Enable FDA Integration on your FDA partner settings, to enable automatic ACK tracking.
In this mode, messages sent to FDA will get tagged with the FDA-assigned
CoreID, and any ACK responses from FDA will also get tagged with the CoreID and ACK number (ACK2, ACK3, etc.).You can filter and track the status of each submission by simply clicking the FDA:CoreID tag on any of the submission entries in your inbox/outbox, or by entering the desired CoreID in the format “FDA:CoreID
" (without <, >) in the search pane.







